 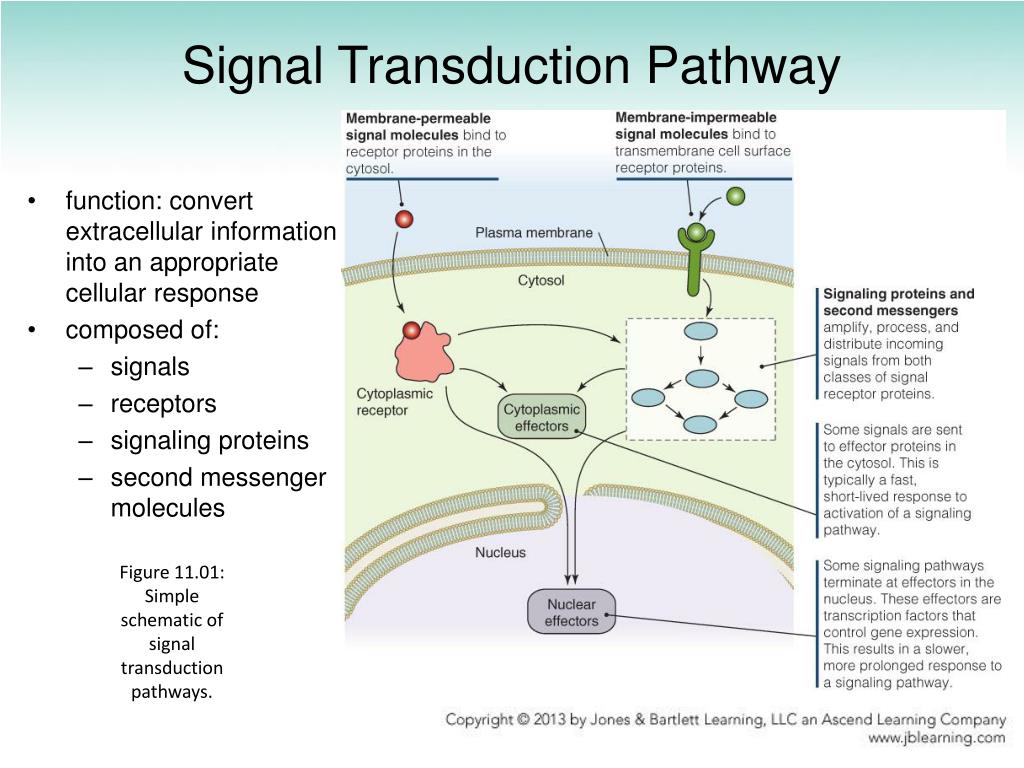
PDB codes are provided in figure, except for NsaS (NMR structure). Structurally elucidated examples include simple coiled-coils (NsaS), HAMP (AF1503), PAS (VicK), GAF (Nlh2), Tandem HAMP (Aer2), and HAMP/PAS domain (VicK). Various protein folds and numbers of signal transduction domains are found inserted between sensor (blue) and autokinase (purple). While some intervening transduction domains have clearly annotated functions, such as the binding of intracellular ligands which are integrated into the sensory function of the HK, the requirement for other signal transduction domains remains obscure. Therefore, they are likely to serve a generalizable function that is robust to evolutionary selection and allow for the construction of physiologically relevant sensors optimally positioned to respond to environmental changes. Moreover, it is clear from the representation of these folds in diverse protein classes that these domains evolved independently of HKs and were incorporated pervasively into functioning HK architectures. It is abundantly clear that the same conserved autokinase domain that defines this protein class can be regulated by a myriad of structural inputs, ranging from short alpha-helical dimeric coiled coils, to well-folded tertiary folds such as HAMP, PAS, and GAF domains ( Krell et al., 2010 Bhate et al., 2015 Figure 1). This question is especially perplexing in light of the various modular architectures of HKs, involving the insertion of one or more signal transduction domains between sensors and the conserved autokinase domain. While these structures offer a conserved view of the catalytic cycle of the cytosolic autokinase domain ( Jacob-Dubuisson et al., 2018), the question of how these proteins couple a sensory event on the other side of the membrane, and many nanometers away to the modulation of the activity of this domain remains unanswered. Several crystallographic snapshots of the autokinase domains of multiple HKs in various conformations ( Ferris et al., 2014 Rivera-Cancel et al., 2014 Wang et al., 2013 Mechaly et al., 2014 Mechaly et al., 2017 Casino et al., 2009 Albanesi et al., 2009), particularly CpxA, DesK, and VicK, have shown distinct conformations involved in autophosphorylation, phosphotransfer, and dephosphorylation that may be conserved across this family. While the lack of a full-length HK structure has hampered our understanding of the mechanism of signal transduction in these proteins, cytoplasmic domain structures have shed light particularly on the enzymatic core of this class of kinases. HKs are constitutive homodimers, which transmit signals through a series of intermediary domains to a cytoplasmic catalytic domain. These sensors are generally implicated in environment sensing and are involved in multi-drug resistance ( Nishino et al., 2005 Hirakawa et al., 2003 Nizet, 2006) and as master regulators of virulence programing in pathogenic bacteria ( Thomas and Cook, 2020 Delauné et al., 2012). Two-component system sensor Histidine Kinases (HKs) are conserved signaling modules in bacteria responsible for sensing a myriad of environmental stimuli and orchestrating transcriptional responses along with their cognate transcription factors (Response Regulators, RR) ( Stock et al., 2000 Groisman and Mouslim, 2006). Our model suggests signals transmit via interdomain allostery rather than propagation of a single concerted conformational change, explaining the diversity of signaling structural transitions observed in individual HK domains. Mutations alter signaling by locally modulating domain intrinsic equilibrium constants and interdomain couplings. The catalytic and sensor domains intrinsically favor a constitutively ‘kinase-on’ conformation, while the HAMP domain favors the ‘off’ state when coupled, they create a bistable system responsive to physiological concentrations of Mg 2+. Data from 35 mutants are explained by a semi-empirical three-domain model in which the sensor, intervening HAMP, and catalytic domains can adopt kinase-promoting or inhibiting conformations that are in allosteric communication. Strikingly, conservative single-site mutations distant from the sensor or catalytic site strongly influence PhoQ’s ligand-sensitivity as well as the magnitude and direction of the signal. 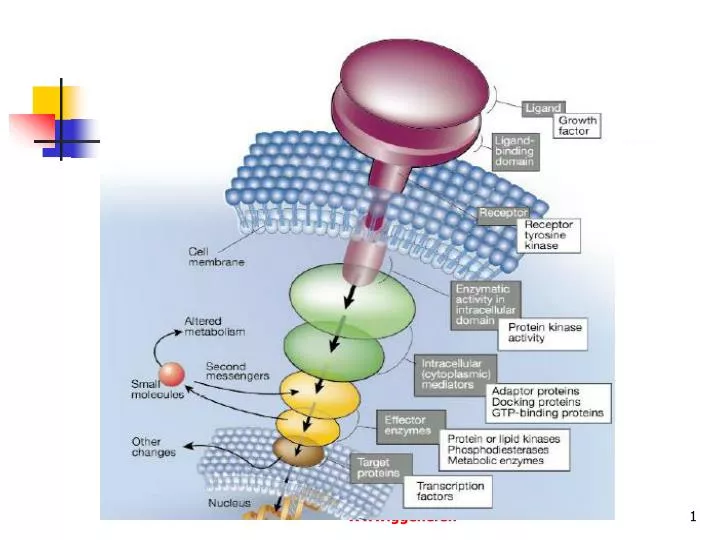
We use cysteine-crosslinking and reporter-gene assays to simultaneously and independently probe the signaling state of PhoQ’s sensor and autokinase domains in a set of over 30 mutants. coli two component histidine kinase (HK), PhoQ, modulates its cytoplasmic kinase domain. Here, we examine how binding of Mg 2+ to the sensor domain of an E. Transmembrane signaling proteins couple extracytosolic sensors to cytosolic effectors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
